Théo Richard, MSc
Aurélien Bustin, PhD
Hubert Cochet, PhD
Victor de Villedon de Naide, MSc
Acute myocardial infarction in the real world
You have probably heard about myocardial infarction (MI), a condition that occurs after ischaemia, i.e. when the blood and oxygen supply to the heart muscle is disrupted. This is the major cause of mortality worldwide, with millions of new cases reported every year (Salari et al., 2023). There are 2 types of MI: chronic MI, which occurred months earlier, and acute MI, which has occurred recently. Acute MI is typically associated with inflammation surrounding the infarcted myocardial tissue. Additional complications may arise, such as papillary muscle infarction (PMI), which concerns small structures inside the heart’s ventricles—the papillary muscles—that are essential in proper cardiac valve function. When these structures are affected, patient outcomes are often worse, and the risk of adverse cardiac events increases significantly.
The reason why it is urgent to quickly treat acute MI, particularly in the presence of PMI, is to limit the risk of potential future major cardiac events. Several imaging techniques can provide critical diagnostic and therapy guidance. Among them, magnetic resonance imaging (MRI) stands out as the reference for myocardial tissue characterisation and precise visualisation of papillary muscle, with the added advantage of being non-invasive and free of ionising radiation.
CMR: a diagnostic treasure trove hard to reach
Cardiac MRI (CMR) is urgently required in every patient with acute MI to assess the extent of damage caused by ischaemia. During the exam, the acute and MI parts of ‘acute MI’ are literally assessed one after the other by 2 different MRI techniques, called sequences: T2 mapping and bright-blood late gadolinium enhancement (LGE), respectively. On the one hand, T2 mapping shows the extent of myocardium that was deprived of oxygen during ischaemia and reperfusion,
called oedema, without differentiating reversibly healthy from permanently damaged areas. On the other hand, bright-blood LGE allows visualisation of the area where ischaemia-induced cell death, called scar, occurs, using a contrast agent injected intravenously into the patient. These 2 biomarkers allow the assessment of reperfusion success and guide therapeutic decisions.
When following this MRI protocol, 2 main issues are encountered:
- Bright-blood LGE may fail to reveal the scar when adjacent to the ventricular blood pool, which can lead to misinterpretation of the real scar extent. This lack of contrast between the blood and enhanced areas concerns, for instance, subendocardial scars (most frequently seen in MI patients) or PMI.
- The injection of a contrast agent creates an interval of at least 20 minutes between the 2 sequences. During this time, the patient may move slightly; in addition, the patient may not hold their breath properly during the scan, leading to different heart positions and unregistered T2-mapping and LGE images, making image analysis difficult.
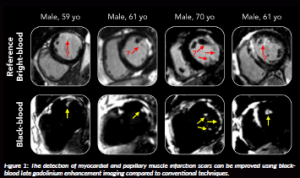
Black-blood LGE represents a solid alternative to bright-blood LGE by totally nulling the blood’s signal in addition to the viable myocardium, thus improving the contrast between scar and blood (Holtackers et al., 2021) (Figure 1).
However, it comes with less anatomical information than bright-blood imaging. The SPOT technology, combining both bright- and black-blood LGE imaging, was recently introduced and validated in over 500 patients in detecting MI (Bustin et al., 2025a).
A novel CMR technology: SPOT-MAPPING
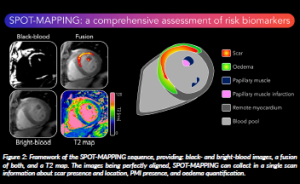
To address the second issue mentioned previously, we developed a CMR technology called SPOT-MAPPING that simultaneously acquires T2 mapping and bright- and black-blood LGE images, with all images co-registered (Bustin et al., 2025b). Because the images are perfectly aligned, the anatomical contours provided by the bright-blood images can be overlaid on the other 2 to optimally extract several useful diagnostic biomarkers. This enables a comprehensive assessment of oedema extent, scar distribution, left ventricular thrombus and PMI presence in a single sequence (Bustin et al., 2026; de Villedon de Naide et al., 2026) (Figure 2).
For this project, we prospectively enrolled 20 acute MI patients undergoing CMR within 4–7 days post-emergent coronary intervention. We showed that SPOT-MAPPING acquisition time (~5 min) reduced by a factor of 2 relative to the conventional acquisition time for both T2 mapping and LGE (~10min). Moreover, SPOT-MAPPING halved manual anatomical contouring (~3min) while maintaining high reproducibility for oedema and scar size. The agreement with conventional bright-blood LGE for scar quantification remained strong. It also improved PMI detection and demonstrated high inter- and intra-observer reproducibility in scar size as a percentage of oedema volume compared with conventional CMR methods.
Papillary muscle infarction: an underdiagnosed biomarker
In the clinic, conventional bright-blood LGE is utilised to assess the presence of a scar within healthy myocardium, but the presence of PMI is often not taken into consideration. Indeed, the latter is often missed due to the lack of contrast between the bright signal of the PMI and the light grey signal of the blood. This represents a real concern, as the outcomes of PMI are known to be dramatic and include mitral regurgitation, larger infarct size, greater left ventricular dysfunction, and, in rare cases, rupture of the PM (Bogun et al., 2008). Properly assessing the presence of PMI is therefore crucial.
Sequences leveraging the benefits of co-registered bright-blood (cardiac anatomy) and black-blood (improved LGE contrast) imaging, such as SPOT or SPOT-MAPPING showed promise in overcoming this issue, by providing improved detection of PMI compared to conventional techniques (Richard et al., 2025). We believe that the clinical integration of such technology would greatly benefit both clinicians and patients alike. Moreover, the improved anatomical information (provided by the bright-blood images) combined with scar and PMI visualisation (provided by the black-blood images) would greatly facilitate the integration of an artificial intelligence (AI)-based analysis, the adoption of which in the clinic has remained mainly compromised to date.
AI-powered CMR exam
Despite being an extraordinary tool, one of the drawbacks of CMR is its complex and operator-dependent analysis. To obtain a precise diagnosis and therapy guidance, medical professionals must perform a meticulous manual analysis of the images collected during the examination for each sequence.
On LGE images, anatomical contours of the heart are delineated in the whole heart. The scar, if present, is segmented as precisely as possible to retrieve its location, volume, transmurality, and to assess what percentage of the heart is damaged. Additionally, PMI presence is visually assessed. On T2 maps, oedema is also quantified and manually segmented. In addition, right ventricular insertion points (corresponding to the intersection between the right and left ventricles) are localised on each image to create a standardised 2D visualisation of the whole heart, called a bullseye. As the images are not registered and are often collected with different imaging parameters (e.g. different field of view, image resolution or number of 2D slices), the heart will not have the exact same spatial location on the images from one sequence to another.
This analysis is time-consuming and can introduce variability, both due to the operator’s expertise and the laborious, repetitive manual tasks they have to perform. Having a fully automated algorithm perform these tasks would allow medical professionals to focus on their expertise and reduce the overall time required for image analysis.
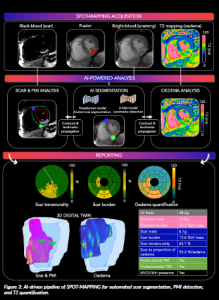
Here, we propose a fully automated processing of the SPOT-MAPPING images (Figure 3):
- A heart segmentation is performed on the bright-blood image using a first transformer-based AI model. A second U-Net-based AI model is used to detect right ventricle insertion points.
- The contours and insertion points are then propagated onto the co-registered black-blood images and T2 maps.
- On the black-blood image, a histogram-based segmentation enables automated scar segmentation and PMI detection.
- On the T2 maps, all the pixel values within the left ventricle are automatically analysed and reported in a 16-segment representation of the heart.
- Finally, a 3D reconstitution of the heart is generated, allowing global visualisation and a comprehensive clinical report.
Future perspectives
Two distinct but complementary directions seem to open for us.
First, we could expand our technologies to a wider spectrum of cardiomyopathies, including ischaemic, non-ischaemic, chronic, or overlapping cardiomyopathies. This project solely focused on patients presenting with acute MI, even though our sequence could benefit other populations, and therefore enable a more comprehensive understanding and diagnosis of cardiovascular diseases overall.
Secondly, with major improvements over the past decade, we believe that AI could further reduce overall acquisition and analysis time when integrated into every step of the cardiac protocol. Our team is already exploring these aspects, including ongoing research on automated black-blood contrast selection (de Villedon de Naide et al., 2024) and an all-in-one software for comprehensive and automated image analysis (He et al., 2025).
It is worth mentioning that the primary goal of AI in CMR is not to replace medical professionals but to assist them in their daily practice, allowing them to focus on their core expertise rather than laborious manual tasks.
From start to finish, CMR is essential in MI patients but remains time-consuming and constraining. Thus, reducing its overall examination time is fundamental. There are still many aspects to be improved to make modern CMR examinations more comfortable, reproducible, and accessible, and we are convinced that AI assistance paves the way in that direction.
References
Bogun, F. et al. (2008) ‘Postinfarction ventricular arrhythmias originating in papillary muscles’, Journal of the American College of Cardiology, 51, pp. 1794–1802. Available at: https://doi.org/10.1016/j.jacc.2008.01.046.
Bustin, A. et al. (2025a) ‘AI-powered SPOT imaging for enhanced myocardial scar detection and quantification’, Nature Communications, 16, 11184. Available at: https://doi.org/10.1038/s41467-025-66166-0.
Bustin A, et al. (2025b) ‘One-click co-registered T2 mapping and dual black-and bright-blood late gadolinium enhancement MRI for comprehensive assessment of myocardial injury after acute STEMI’, European Heart Journal Imaging Methods and Practice, 3(2), qyaf103. Available at: https://doi.org/10.1093/ehjimp/qyaf103.
Bustin, A. et al. (2026) ‘Left Ventricular Thrombus Assessment Using Joint Bright- and Black-blood LGE Imaging’, Journal of Cardiovascular Magnetic Resonance, 28, 102218. Available at: https://doi.org/10.1016/j. jocmr.2025.102218.
de Villedon de Naide, V. et al. (2024) ‘Fully automated contrast selection of joint bright- and black-blood late gadolinium enhancement imaging for robust myocardial scar assessment’, Magnetic Resonance Imaging, 109, pp. 256– 263. Available at: https://doi.org/10.1016/j.mri.2024.03.035.
de Villedon de Naide, V. et al. (2026) ‘Clinical applications of co-registered myocardial T2 mapping and dual bright- and black-blood late gadolinium enhancement magnetic resonance imaging’, European Heart Journal – Imaging Methods and Practice, 4(1), qyag030. Available at: https://doi.org/10.1093/ehjimp/qyag030.
He, K. et al. (2025) ‘SPOTCardio: integrated application for AI-powered automated myocardial scar quantification on joint bright- and black-blood late gadolinium enhancement MRI images’, Journal of Clinical Medicine, 14, 8428. Available at: https://doi.org/10.3390/jcm14238428.
Holtackers, R.J. et al. (2021) ‘Dark-blood late gadolinium enhancement cardiovascular magnetic resonance for improved detection of subendocardial scar: a review of current techniques’, Journal of Cardiovascular Magnetic Resonance, 23, 96. Available at: https://doi.org/10.1186/s12968-021-00777-6.
Richard, T. et al. (2025) ‘Improved and automated detection of papillary muscle infarction using joint bright- and black-blood late gadolinium enhancement MRI’, Journal of Magnetic Resonance Imaging, 62, pp. 827–839. Available at: https://doi.org/10.1002/jmri.29777.
Salari, N. et al. (2023) ‘The global prevalence of myocardial infarction: a systematic review and meta-analysis’, BMC Cardiovascular Disorders, 23, 206. Available at: https://doi.org/10.1186/s12872-023-03231-w.
PROJECT SUMMARY
Acute myocardial infarction (MI) is the major cause of mortality, with millions of cases worldwide. Papillary muscles, which are essential in the correct functioning of the heart, can also be infarcted as a complication of MI, which involves even worse outcomes. Cardiac MRI (CMR) is the reference imaging for a radiation-free assessment of the whole heart. However, many individual sequences need to be played out to assess all the relevant biomarkers of the heart, and a thorough analysis of the collected images is required for proper diagnosis and management of patients. This cardiac protocol is long and complex due to the numerous sequences, and the analysis process is equally difficult due to the repetitive and operator-dependent manual tasks. The aim of this project is to (i) propose a sequence called SPOT-MAPPING, that will collect crucial imaging biomarkers in a single scan, and (ii) fully automate analysis of the obtained images. We believe that this approach is ideal for patients presenting with acute MI and papillary muscle infarction.
PROJECT LEAD PROFILE
Aurélien Bustin is an Assistant Professor at IHU LIRYC, the Heart Rhythm Disease Institute, at Bordeaux University (France) and a visiting researcher at the Department of Diagnostic and Interventional Radiology, Lausanne University Hospital and the University of Lausanne (Switzerland) since 2020. Dr Bustin is the project lead for SMHEART, which started in September 2023. His team publishes within the fields of radiology, cardiac magnetic resonance imaging, and computer science.
PROJECT CONTACTS
Prof. Jr. Aurélien Bustin
IHU LIRYC, Heart Rhythm Disease Institute,
Hôpital Xavier Arnozan, Université de Bordeaux – INSERM U 1045 Avenue du Haut Lévêque, 33604 Pessac, France
Email:aurelien.bustin@ihu-liryc.fr
Web: smheart.eu
LinkedIn: /in/aurelien-bustin
LinkedIn: /company/project-smheart-ihu-liryc
FUNDING
This research was supported by funding from the French National Research Agency under grant agreements Equipex MUSIC ANR-11-EQPX-0030, ANR-22- CPJ2-0009-01, and Programme d’Investissements d’Avenir ANR-10-IAHU04-LIRYC, and from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (Grant agreement No. 101076351).
Figure legends
Figure 1: The detection of myocardial and papillary muscle infarction scars can be improved using black-blood late gadolinium enhancement imaging compared to conventional techniques.
Figure 2: Framework of the SPOT-MAPPING sequence, providing: black- and bright-blood images, a fusion of both, and a T2 map. The images being perfectly aligned, SPOT-MAPPING can collect in a single scan information about scar presence and location, PMI presence, and oedema quantification.
Figure 3: AI-driven pipeline of SPOT-MAPPING for automated scar segmentation, PMI detection, and T2 quantification.